Susan L. Clubb, DVM
Loxahatchee, Florida
Alan Herron
Comparative Pathology Laboratory
University of Miami Miami, Florida
Session # 2020
Summary: Feather discoloration in psittacines has been attributed to aging of feathers, delayed moult, malnutrition, hypothyroidism, and often to liver disease.1 Feather discoloration due to soiling and subsequent saprophytic fungal growth has been described in flamingoes at Slimbridge England.2
Feather Fungus in Flamingoes
This phenomenon was also observed at Parrot Jungle and Gardens in Miami after a dietary change, which increased the fat content of the diet. The flock of 57 Caribbean flamingoes was housed in a large open-air exhibit approximately 1 acre, with a large fresh water lake. The birds remained almost exclusively in the open sunny center of the exhibit adjoining the lake. The exhibit was surrounded by a well maintained but dense garden.
Addition of a high quality dog food to the diet was intended to improve reproductive performance in the stable flock of flamingoes. The dietary change was followed by onset of the rainy season. Flamingoes engaged in nest building activities also tend to soil their plumage with mud used to construct mounds. The plumage, primarily of the back and wings, developed a dark discoloration appearing streaked with black. Physical exam revealed oily deposits coating the feathers with grossly visible opaque black coloration. Microscopic examination revealed black deposits on plumules often with corresponding degeneration of pennaceous barbs and barbules.
Culture of feathers revealed several species of saprophytic fungi which are also associated with sooty mold, a fungal problem of plants associated with aphid infestation. Secretion of a nutritious excrement by aphids encourages saprophytic fungal growth.
Capture and cleansing of all birds in the flock was considered impractical and potentially harmful. Palliative actions included dietary alteration and attempts to alter the progression of the problem. The flamingoes diet is provided in a soaked mixture. The oil from the dog food was floating on the surface and coating the beaks of the flamingoes. An inexpensive dog food was selected based subjectively on the amount of oil, which floated to the surface after mixing the food in water. This was determined by dipping hands into bowls of soaked dog food. This simulated oil deposition on the face of the flamingoes during feeding which was subsequently rubbed on plumage of the back and wings during preening and rest. Chlorhexadine was also added to the food for antifungal activity. Normal molting and reduced deposition of oily substances allowed natural resolution of the problem within several months.
Control measures also included inspection of surrounding vegetation and spraying with insecticides as necessary to reduce environmental mold exposure.
Feather Fungus in Psittacines
Black deposits on plumage are frequently observed in psittacines. Subjective observations indicate a higher incidence in pet birds housed indoors as compared to avicultural birds housed outdoors. Pet birds that are frequently handled seem to have an increased incidence. As in flamingoes observations indicate that oily deposits on feathers provide a substrate for saprophytic fungal growth. Sources of oil include oil from the hands or face of owners in birds that are frequently handled. Deposits in psittacines also tend to appear on the covert feathers of the back and wings, also indicating that these feathers are used to cleanse the beak after eating or soiled during preening or sleeping or by dunking foods in the water. In macaws deposits are often seen on the more anterior coverts of the wings which may correspond to soiling by food.
Damaged feathers, such as coverts and retrices which are barbered- or feathers which display stress related malformations such as stress lines/bars also seem to be more commonly affected. Wetting of feathers such as frequent bathing, or dipping the tail in water bowls may also enhance growth.
Feathers from ten different birds were examined histologically using routine (H&E) staining. The radius, barbs, and barbules had a mild to moderate growth of mostly round, darkly pigmented fungus. The feather shafts were not affected. Gomori methenamine silver (GMS) stained tissues were consistent with a mixed fungal infection. The results of fungal isolation cultures of affected feathers are presented in Table 1.
Cultures of normal feathers (no visible discoloration) also indicate the presence of saprophytic fungi, but more sporadic and in much lower relative concentrations, indicating that without a build-up of substrate, the fungal growth does not become problematic. A comparison of results of fungal isolation on normal feathers (no visible indication of saprophytic fungal colonization) is presented in Table 2.
Control measures included removal of badly soiled feathers and topical application of chlorhexadine. Under isoflurane anesthesia heavily soiled covert feathers are plucked. Plucking retrices is avoided due to the potential for follicular damage. In the authors opinion removal of heavily soiled feathers results in a more rapid resolution of the problem. Following removal of heavily soiled feathers; birds are misted with a 10% dilution of chlorhexidine solution once or twice weekly until feathers have regrown. Alternative therapies are being investigated. Husbandry and hygiene issues must also be addressed.
Discussion
While primarily a cosmetic problem, a sprophytic fungal growth on feathers may also present a health risk. The most common contaminant is Aspergillus sp., and certainly sleeping with the face embedded in feathers contaminated/encrusted with aspergillus represents an increased risk of systemic aspergillosis.
Subjectively, this feather disorder does appear to be more common on birds in poor or marginal health as opposed to vibrant health. Poor hygiene maybe contributory. It also appears to predominantly affect green and blue feathers (excluding flamingoes), although this many be due to a predominance of green and blue birds examined.
A review of the literature regarding the significance of black discoloration of feathers indicated a supposition that liver disease should be implicated.3 This may lead to a misinterpretation of clinical signs. Other causes of feather discoloration are also poorly documented.1 In a recent publication lipophilic yeasts were described as emerging nosocomial pathogens in a nursery for human infants.4
Primary systemic disease is unlikely as a direct cause as deposits are not uniform as would occur with systemic disease affecting feather growth. The entire feather is rarely affected. Deposits tend to occur on the exposed tips of feathers and not on the protected surfaces which underlie other feathers. Degeneration of the plumules, barbules etc. may be mechanical or due to keratinolytic metabolites of the fungi.
Conclusion
In this report, black deposits on feathers were a multifactoral problem involving deposition of a nutrient source on feathers, colonization of saprophytic fungi, and enhancement by moisture.
In assessment and control of feather abnormalities, which include saprophytic fungal growth, all of these factors including the behavior of both the bird and the owner, must be considered.
References
1. Cooper JE, Harrison GJ. Dermatology. In: Ritchie BW, Harrison GJ, Harrison LR (eds). Avian medicine: principles and application. Lake Worth, FL: Wingers Publishing Inc, 1994: 607-639.
2. Kear J, Deplaix-Hall N. Flamingoes, T. and A.D. Poyser, Ltd., 281 High St. Berkhamsted, Hertfordshire, England, Wildfowl Trust,1975.
3. Lucas AM, Stettenheim PR, Avian anatomy, integument, Volume I, Agricultural Research Service, United States Dept of Agriculture, Agriculture Handbook 362, 1972.
4. Chang HJ, Miller H, Watkins N, et al. An epidemic of malassezia pachydermatis in an intensive care nursery associated with colonization of health care workers pet dogs. New Eng J Med 1998; 338(11):706-711.
Table 1.
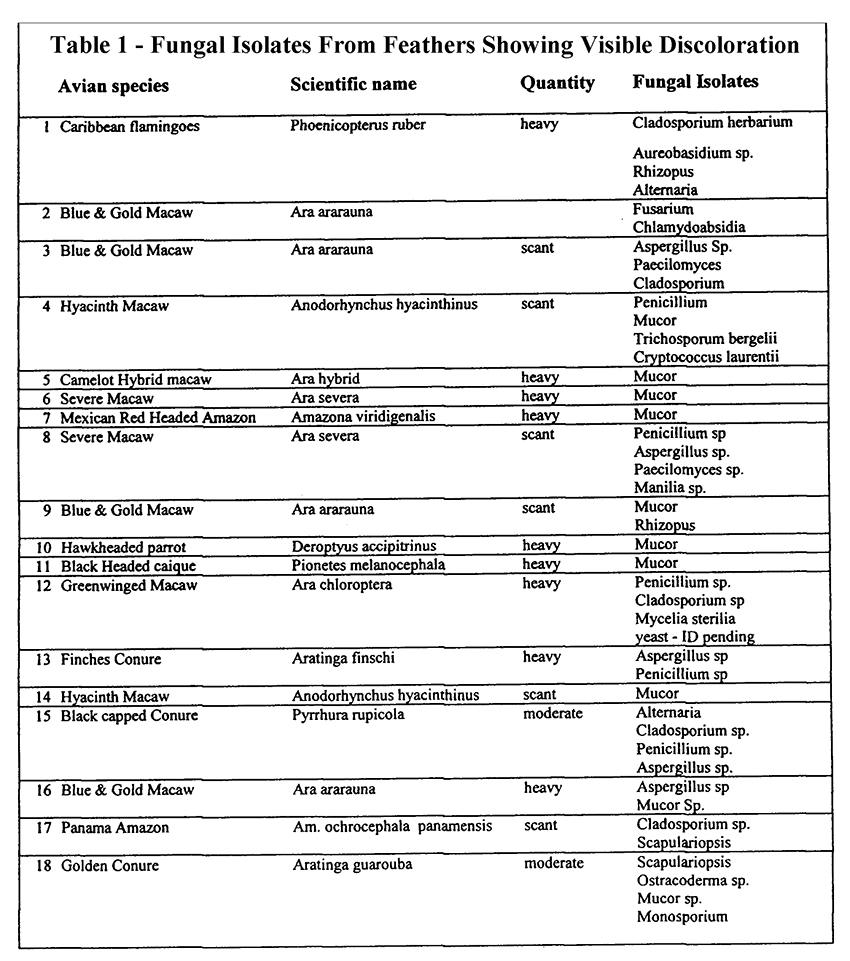
Table 2.

