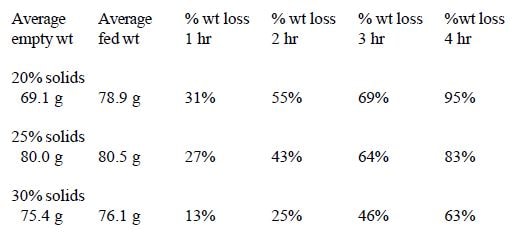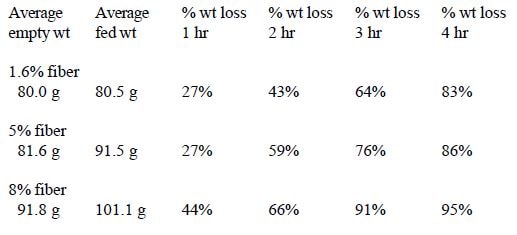Susan L. Clubb, DVM
Michael Massie
Everett Butler
Loxahatchee, Florida
Session # 800
Financial support for this study was provided by Pretty Bird International, Stacy, Minnesota.
Intestinal stasis, leading to crop stasis and souring, is a common sequela of many disease and management problems in neonatal psittacines. Clinical observations have indicated that a number of physical and dietary variables can influence intestinal transit time in psittacine chicks, including percentage of solids, fat content, protein content, temperature of the formula, and temperature of the chick’s environment.4,5
Many disease processes can result in delayed intestinal transit time in handfed psittacine chicks, including bacterial, viral, and fungal infections. Dehydration and absorption of fluids from handfeeding formulas can potentiate these effects. Veterinarians typically treat such ill chicks with oral or parenteral fluid therapy or diluted formulas, which, if continued for more than a few days, can result in inadequate caloric intake and deficiencies of some nutrients.
For handrearing techniques to be most successful, a formula must provide adequate nutrition and move through the gastrointestinal system at a rate which will maximize absorption and utilization of nutrients without slowing transit time, encouraging the proliferation of pathogenic bacteria. Control of intestinal transit time can also be useful in designing a formula which allows the caretaker to feed on a reasonable schedule. The manipulation of types and quantities of fiber products in handrearing formulas may be used to regulate intestinal transit time. Before this can be accomplished, however, the variables affecting intestinal transit time must be known.
This study was designed to preliminarily assess the effects of some dietary parameters on intestinal transit time in handfed psittacine chicks, and to indicate areas needing more exhaustive analysis.
Materials and Methods
Normal, clinically healthy cockatiel (Nymphicus hollandicus) and quaker parakeet (Myopsitta monachus) chicks were used as experimental subjects. All chicks had been parent-raised for at least 1 week prior to removal from the nest for handfeeding. Chicks were randomly placed in groups with sizes matched as closely as possible. Individual chicks were identified by closed leg bands. Chicks were fed 4 times daily, and all had been removed from the nest at least 4 days prior to the trial. All chicks were fed a commercially formulated diet containing 19% protein and 12% fat (19/12 Handrearing Formula—Pretty Bird International, Stacy MN). This formula was used as the standard or control formula for the feeding trials. All trials were performed in the afternoon after chicks had been fed normally in the morning.
For each study, chicks were weighed prior to feeding, fed a measured amount of the experimental diet, and weighed again to determine the weight gain. Chicks were then weighed each hour for four hours in order to determine how rapidly they lost the weight gained in the feeding.
Trial # 1
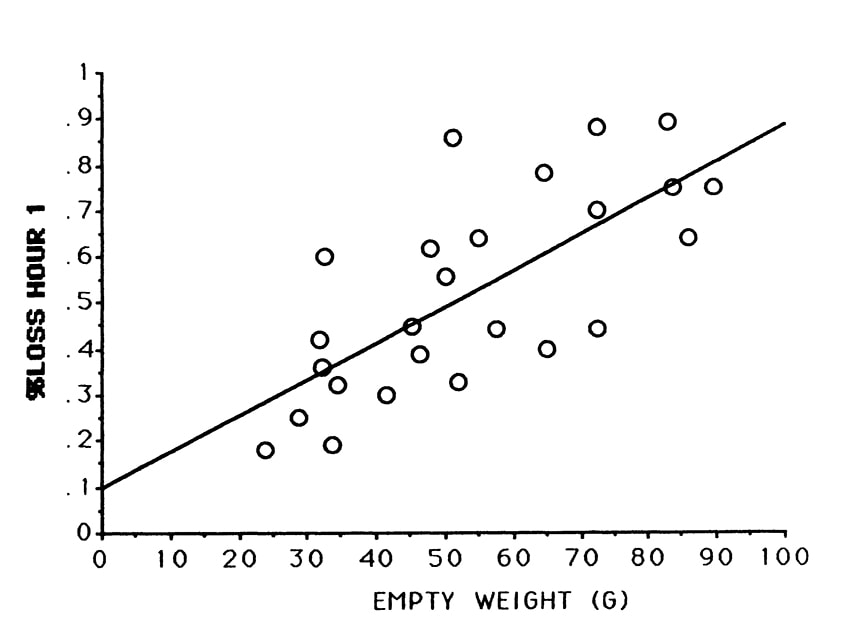
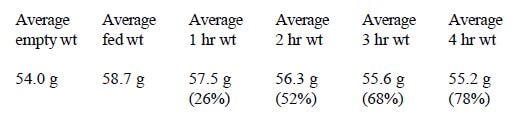
Twenty-five (25) quaker parakeet chicks varying in size from 24 grams to 89.5 grams were fed roughly the same volume (5 ml) of food. The smallest birds could not hold the full volume and were fed until the crop was full. The standard formula was used, mixed at 25% solids. This study addressed the relationship between chick size and transit time.
Trial # 2
Twenty-five (25) quaker parakeet chicks varying in size from 46 to 120 grams were fed 10% of their body weight in formula. The standard formula was used, mixed at 25% solids. This study examined the size/time relationship with volume of food proportional to size.
Trial # 3
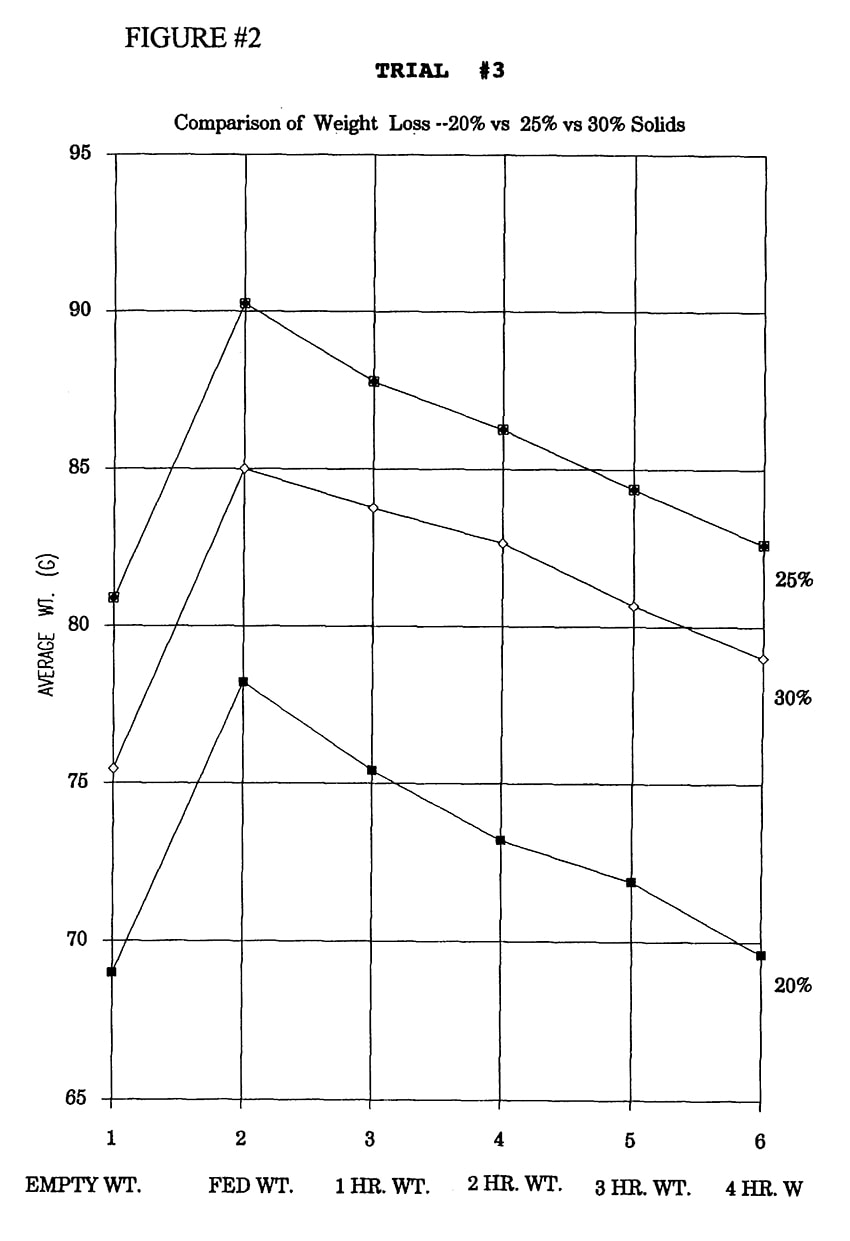
The standard formula was mixed at three different concentrations-20%, 25% and 30% solids. Ten mls. were fed to cockatiel chicks in three study groups of 10 birds each.
Trial # 4
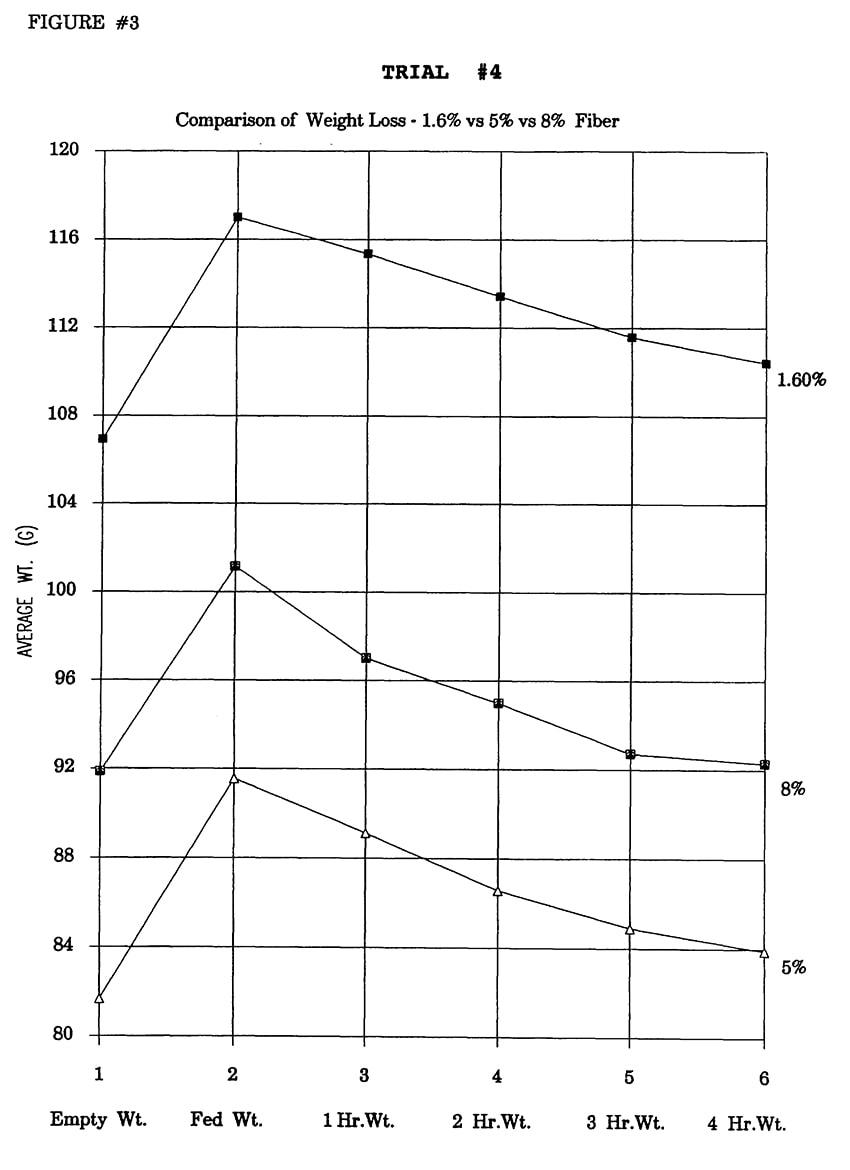
In Trial #4, the effects of dietary fiber level were examined. The standard formula, which is 1.6% fiber, was used as a base formula for a trial designed to determine the effects of insoluble fiber on intestinal transit time. Soyhulls were used to increase the fiber level of the diet to 5% and 8%. Soyhulls were ground to a fine particle size in a blender. Potato protein and 760 fat (7% protein, 60% fat product consisting of coconut oil and animal fat) were also added in the proper proportions to maintain the protein at 19% and fat at 12%.
Trial # 5
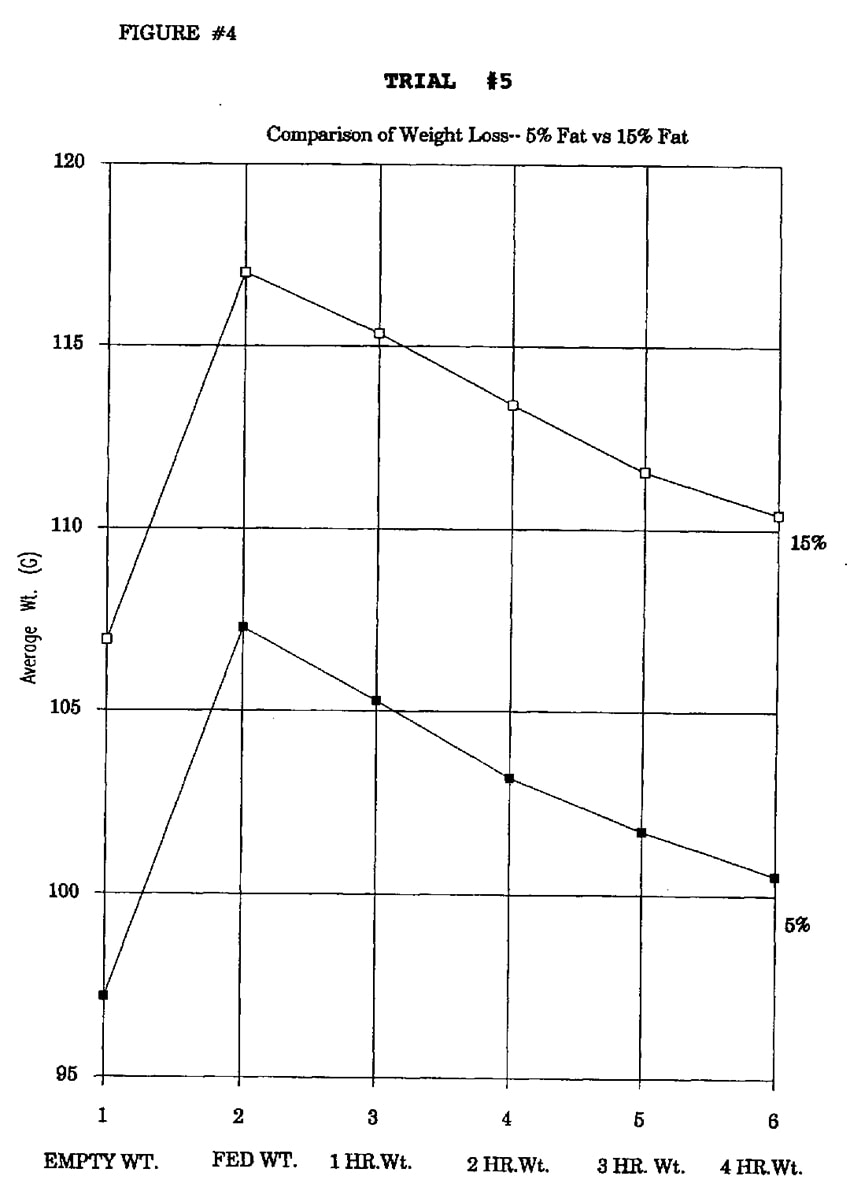
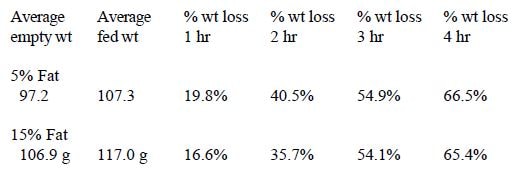
In Trial #5, the effect of dietary fat level was examined. Two formulas which varied only in fat and carbohydrate levels were compared. Formula #1 contained 19% protein and 5% fat, and formula #2 contained 19% protein and 15% fat. Both formulas contained 1.6% fiber.
Results were analyzed using Statview software for the Macintosh (Statview SE+ Graphics, Abacus Concepts, Berkeley CA). One and two hour data were selected for analysis in Trials #3 and #4, because no birds in these groups had lost 100% of the weight gained in feeding. All percent loss data at 1 and 2 hours were arc-sin transformed prior to analysis by 1-way ANOVAS or unpaired t-tests.
Results
In Trial #1, a significant correlation occurred between size and intestinal transit time. Smaller chicks emptied more slowly. This may have occurred because the crops of smaller chicks were completely filled while the crops of the largest chicks were only half filled (Figures 1 & 2).
In Trial #2, chicks which were fed volumes of formula proportionate to their size (10% of body weight) appeared to empty at generally the same rate; however, since food volume and clutch size both varied, percent weight loss may not actually be independent of chick size. These data should be interpreted with caution.
In Trial #3, significant differences in weight loss occurred among groups fed different percent solids at 1 hour (1- way ANOVA, df=2,24, p=.0012) and 2 hours (1-way ANOVA, df=2,24, p=.0001). The 20% and 25% solids treatment groups were both significantly different from the 30% solids treatment group (Fisher PLSD, p <.05) at 1 hour, and all three treatment groups were significantly different from each other (Fisher PLSD, p<.05) at 2 hours (Figure 3).
In Trial #4, significant differences in weight loss occurred between groups fed different fiber levels at 1 hour (1- way ANOVA, df=2,20, p=.0039) and 2 hours (1-way ANOVA, df=2,20, p=.0056). The 1.6% and 5% fibers were significantly different from 8% respectively at both 1 and 2 hours (Fisher PLSD, p<.05). No significant differences were found between 1.6% and 5% fiber treatment groups (Figure 4).
In Trial #5, no significant difference in weight loss occurred between the 5% and 15% fat treatment groups at 1, 2, 3, or 4 hours (Figure 5).
Conclusion
In these trials, several dietary variables had a significant influence on intestinal transit time in chicks. A significant correlation was seen between transit time and size, with smaller chicks emptying more slowly. As illustrated in Trial #2, however, this difference may also be related to fullness of the crop. Transit time was affected by percent solids of the formula fed being significantly slower in chicks fed 30% solids as opposed to 20% or 25% solids. Percentage of insoluble fiber in the diet also affected transit time. A significant increase in transit time occurred when the diet contained 8% insoluble fiber as opposed to 5% and 1.6%. Percent fat did not affect gut transit time.
Discussion
In handfed psittacines, regulation of intestinal transit time is important for a number of reasons. Rapid gut transit time could result in poor digestion and absorption of nutrients leaving chicks hungry, begging, and potentially malnourished unless fed very frequently. Rapid intestinal transit minimizes the normal absorption of ions and water through the colonic mucosa, thus contributing to increased fecal water content. It also strains the handfeeder with more frequent feedings. Conversely, very slow gut transit time can result in bacterial overgrowth and souring of food in the crop and intestinal tract. A happy medium can provide maximum nutrition for the chick and a livable feeding schedule for the handfeeder.
Dietary fiber has traditionally been considered an inert component of foods and used to regulate intestinal transit time and consistency of feces. The first use of fiber as a laxative is attributed to Hippocrates.1 Botanically, fiber refers to the polysaccharide and lignin portions of the plant cell wall. Dietary fiber can be grouped into two broad classifications—soluble and insoluble—according to fiber type and physiologic effects. Soluble fibers (pectins, gums, and some hemicelluloses) have great water-holding capacities, delay gastrointestinal transit time, and are completely fermented by colonic microflora. Soluble fibers can absorb a large quantity of water and form gels.1,2
In contrast, insoluble fibers (cellulose, lignin, and some hemicelluloses) have less water absorptive capacity, hasten gastrointestinal transit time, and are less efficiently degraded by colonic microflora.3 The effects of insoluble fiber on gastrointestinal transit time, fecal bulking, and water absorption make insoluble fiber more helpful than soluble fiber in the treatment of constipation in mammals. Insoluble fiber is also capable of retaining water within their fiber matrix. The particle size of a fiber affects its water-holding capacity; courser particles retain greater quantities of water. Increased fecal bulk and weight stimulates the defecation reflex and shortens intestinal transit time.3 Psyllium seed hydrocolloid (Metamucil, Searle Consumer Products) contains hemicelluloses that have properties of both insoluble and soluble fiber. Understanding the effects of various forms of fiber on gut transit time in handfed psittacines could be useful in designing hand feeding formulas which would be evacuated by chicks at a predictable and/or controllable rate.
These trials are preliminary in a field which needs further investigation. A potential problem in the experimental design was that chicks of uniform size were not available, as chicks were rapidly growing and changed in weight daily. The relationship of percent solids and the number of variables in fiber sources alone—the type and solubility of the fiber source, the effects of fiber particle size and the relationship between fiber digestibility and intestinal microflora—are all areas which warrant further study.
References
1. Eastwood MA. Fiber in the gastrointestinal tract. Amer J Clin Nutr 1978;31:S30-S32.
2. Dimski D. Using dietary fiber to manage constipation. Columbus, OH: Department of Veterinary Clinical Sciences, College of Veterinary Medicine, Ohio State University.
3. Burrows CF, et al. Effects of fiber on digestibility and transit time in dogs. J Nutr 1982;112:1726-1732.
4. Voren H, Jordan R. Parrots—handfeeding and nursery management. Ontario, Canada: Silvio Mattacchione & Co., Pickering, 1992.
5. Clubb SL, Wolf S, Phillips A. Psittacine pediatric medicine. In: Psittacine Aviculture: Perspectives, Techniques and Research. Loxahatchee, FL: Avicultural Breeding and Research Center, 1992.
FIGURE #1
TRIAL #1
Relationship Between Chick Size and Transit Time (Chicks Fed Same Volume Formula)