Susan L. Clubb, DVM, Dipl ABVP (Avian), and Siwo De Kloet, PhD
Session #205
Affiliation: From Rainforest Clinic for Birds and Exotics, 3319 E Rd, Loxahatchee, FL 33470, USA (Clubb) and Animal Genetics Inc, 1336 Timberlake Rd, Tallahassee, FL 32312, USA (De Kloet).
Abstract: A battery of diagnostic tests including crop biopsy, serology by enzyme-linked immunosorbent assay, and reverse transcription polymerase chain reaction to detect avian bornavirus antigen in choanal and cloacal swabs was used to screen asymptomatic psittacine birds in 4 collections where clinical cases of proventricular dilatation disease had previously occurred.
Proventricular dilation disease (PDD) is a fatal inflammatory disease affecting primarily psittacine birds (Order Psittaciformes). In its classic form, PDD is a wasting disease characterized by weight loss, maldigestion, and central nervous disorders. PDD has been reported in over 70 psittacine species. In 2008, Kistler et al and Hanka- vuori et al independently reported on the recovery of a novel bornavirus, designated avian bornavirus (ABV), in birds with PDD from the US and Israel using advanced molecular tools such as panviral DNA microarrays and high throughput sequencing.1,2 PDD has been successfully reproduced in cockatiels (Nymphicus hollandicus) inoculated with brain homogenate containing ABV 4, and the presence of an ABV 4 nearly identical to that of the inoculum was demonstrated in various organs of the inoculees.3 PDD has also been reproduced in cockatiels and Patagonian conures (Cyanoliseus patagonus) using cultured ABV, fulfilling Koch’s postulates.4
PDD is often found as a disease of low incidence in avicultural collections. The presence of pathologic lesions of PDD, as determined by crop biopsy as well as the detection of ABV in asymptomatic birds has also been reported.4–6 The existence of asymptomatic or latent ABV infections complicates attempts to manage avicultural or companion psittacine collections and screening of contact birds after the diagnosis of PDD in a facility. Select- ing the correct test, or battery of tests, is an important aspect of management of subclinical infections in building confidence in the disease status of birds being offered for sale, or new birds entering an established aviary.
In this study, 137 birds in 4 aviaries where PDD had previously been were screened in an attempt to determine the health status and presence of PDD or ABV infection in the aviaries.
Materials and Methods
Birds utilized in this study are client-owned. In each collection, there had previously been at least one death due to PDD. Three collections are housed outdoors in Florida, 2 collections in suspended pair cages, and 1 collection housed in large outdoor flights that are adjacent and share a single concrete floor. These 3 collections are fed a diet composed of extruded formulated diets with a seed mix and vegetables. The fourth collection is housed in an indoor aviary and fed an extruded formulated diet.
In each collection, birds were not showing overt clinical signs of PDD, but were exposed to diseased birds. Some birds were mildly underweight or unthrifty but would not be considered clinically ill. All birds were screened by crop biopsy, serology by enzyme-linked immunosorbent assay (ELISA), and shedding of ABV by reverse transcription polymerase chain reaction (rtPCR).
All samples were collected at each aviary utilizing portable anesthetic and surgical equipment. Under isoflurane anesthesia, skin over the crop area was aseptically prepared and a midline skin incision made, exposing the crop. A section of crop including a prominent blood vessel was collected and crop and skin were closed routinely. Blood was collected from the brachial vein while under anesthesia and placed in lithium heparin tubes for ELISA testing. These samples were placed immediately in a cooler on ice and centrifuged separating plasma using standard techniques. The oropharynx, then the cloacal mucosa, were swabbed and the swab was submerged in solution to stabilize the viral RNA (0.5 ml of 4M guanidinium isothiocyanate, containing 50 mM Tris-HCL buffer [pH 7.5] and 25 mm ethylene diamine tetra-acetic acid [EDTA]). This solution will reportedly stabilize the ABV RNA for 2 weeks and shipping on ice is not necessary (S. R. K., unpublished data). Plasma and swab samples were stored under refrigeration and shipped to the lab overnight.
Pathology was performed by the University of Georgia pathology department and classified as positive (pres- ence of lymphoplasmacytic ganglioneuritis consistent with PDD); not detected if lesions compatible with PDD were not detected in 3 or more ganglia; suspicious if changes highly suggestive of PDD were present but not definitive; or insufficient if less than 3 ganglia were present in the section. Some examples of suspicious diag- nosis would be hypercellular ganglia and nerves, perivascular lymphocytes and plasma cells, or perivascular and perineural mast cells.
The rtPCR analysis used to detect bornavirus was performed using consensus primers described by Kistler et al.1 For ELISA testing, the detection of ABV P40-specific antibodies was carried out as previously described by Kloet and Dorrestein.5 Synthesis of recombinant bornavirus P40 (N) was synthesized in Escherichia coli. Bornavirus P40 nucleoprotein was selected because it is a major bornaviral antigen in mammals.
Results
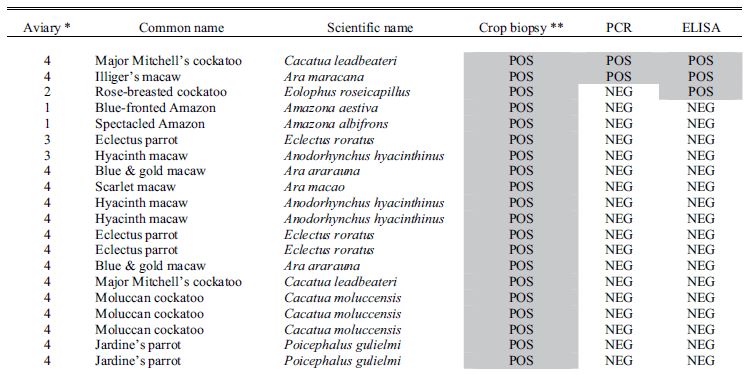
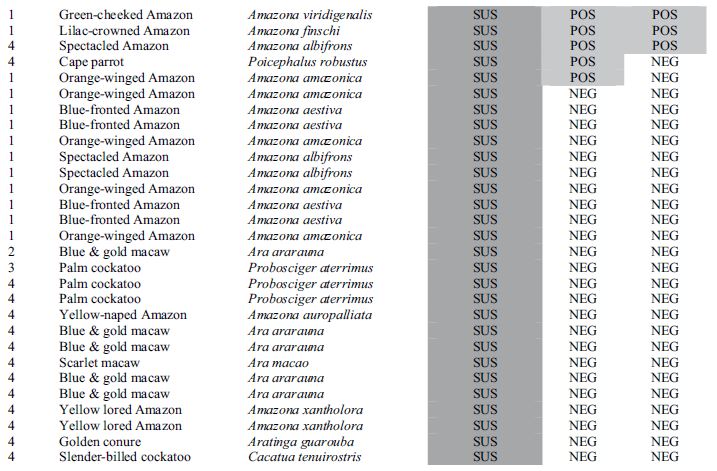
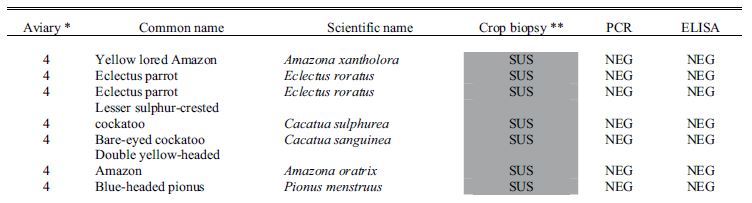
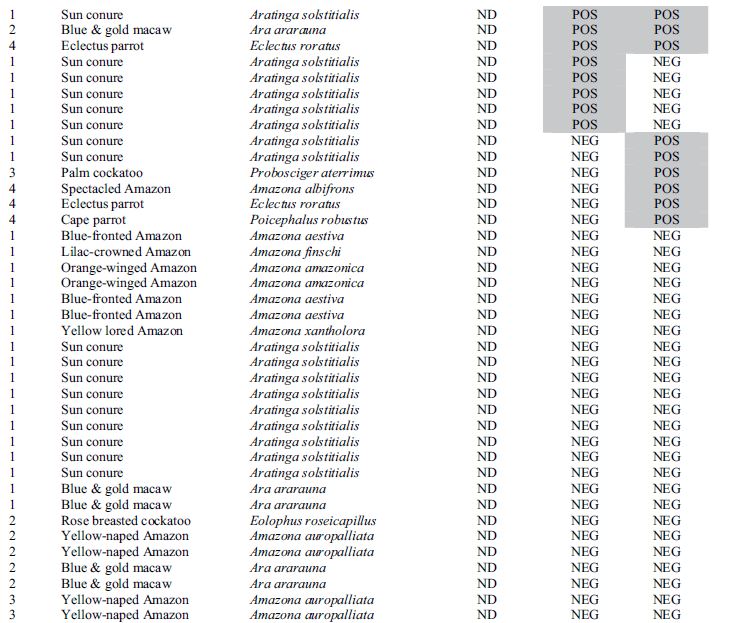
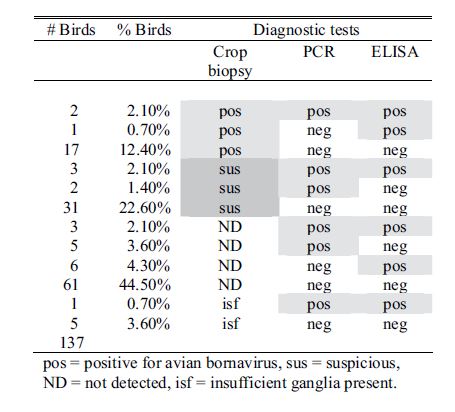
Results for all 4 aviaries are combined and presented in Table 1. There seems to be poor correlation between crop biopsy, ELISA testing, and ABV detection by PCR, as is summarized in Table 2. Of the 137 birds, crop biopsy results are as follows: 20 birds (14.5%) positive, 36 (26.2%) suspicious, 75 (54.7%) not detected, and 6 insufficient ganglia to make a diagnosis. ELISA testing identified 16 birds (11.6%) positive for ABV P40-specific antibodies and had relatively low correlation with crop biopsy results. The rtPCR testing identified 9 (6.6%) of the birds to be shedding the virus at the time of sample collection positive, and this test also did not correlate well with crop biopsy results.
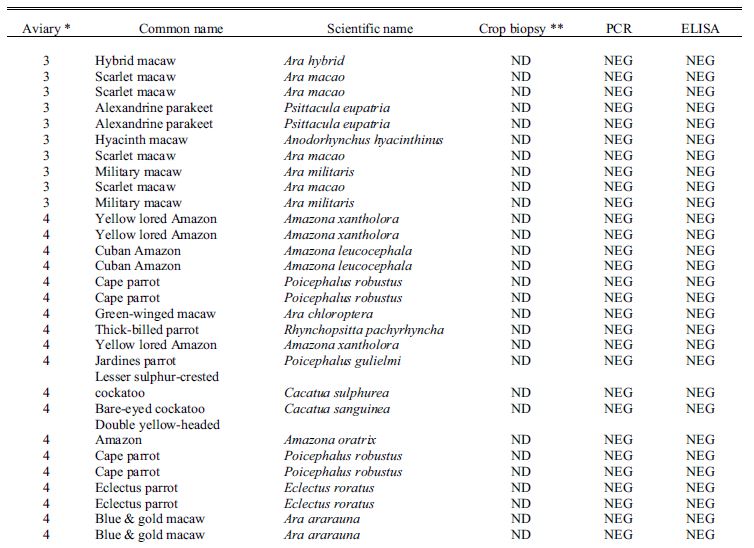
Table 1. Results for diagnostic tests including crop biopsy, serology by enzyme-linked immunosorbent assay, and reverse transcription polymerase chain reaction used to detect avian bornavirus antigen in choanal and cloacal swabs used to screen asymptomatic psittacine birds in 4 collections where clinical cases of proventricular dilatation disease had previously occurred.
* Aviary 1 was indoors; Aviaries 2 and 4 were outdoor suspended cages; while aviary 3 had flights on a concrete slab.
**Crop biopsy key: POS = positive, ND = not detected, SUS = suspicious, ISF = insufficient ganglia present.
Table 2. Summary results for identification of avian bornavirus in 137 birds from 4 aviaries as determined by crop biopsy, PCR, and ELISA. Note poor apparent correlation between results from each method.
pos = positive for avian bornavirus, sus = suspicious, ND = not detected, isf = insufficient ganglia present.
Discussion
Detection of lymphoplasmacytic ganglioneuritis by histology has long been considered the “gold standard” for diagnosis of PDD. With the discovery of ABV and the positive correlation of ABV to PDD, the development of non-invasive diagnostic screening tests ensued in a quest to provide practical, non-invasive screening of populations for the disorder. Several issues should be considered in this study. The potential that another agent might cause lymphoplasmacytic ganglioneuritis cannot be excluded. Tissue samples submitted to the Univer- sity of Georgia were not directly tested by PCR or immunohistochemistry for the presence of ABV. The direct relationship between lymphoplasmacytic ganglioneuritis found on crop biopsy and the classic clinical form of PDD requires further investigation. Additionally, ABV has been reported in clinically healthy birds, further complicating the clinical picture.5-7
RtPCR assays for ABV from cloacal swabs are generally less sensitive assays due to intermittent shedding of ABV, which has been documented, or the possibility that viral RNA was degraded due to poor handling procedures.8 Biological explanations for the discrepancy might include the existence of birds that are not shedding because their intestinal tracts are not affected, or alternatively individuals in which the infection is transient, establishing an inflammatory process that becomes latent, much as an autoimmune disease.
The low rate of positives on ELISA testing was more puzzling. It must be noted that only P40 ABV specific antigen was used for ELISA testing during this study. While the P40 protein has been used extensively in mammalian bornavirus serology, it is possible that other antigens including P29, P24, and matrix protein used in current ABV panels may identify additional birds that have been immunologically exposed to a specific ABV antigen. This study also shows that more work must be done to explain causes of inflammation in birds. Work is currently being conducted to develop a method of accurately measuring systemic inflammatory factors including cytokines like tumor necrosis factor-alpha. These serological assays could play a role in better understanding and more accurately identifying certain inflammatory factors that stimulate an acute phase reaction. Additionally testing for alpha and gamma interferon levels may lead to more comprehensive understanding of the immune systems response to these infectious agents. The pathogenesis of avian bornavirus infections is still poorly understood. Triggers that exacerbate an asymptomatic infection resulting in clinical PDD are likewise poorly understood.
References
1. Kistler AL, Gancz A, Clubb S, et al. Recovery of divergent avian bornaviruses from cases of proventricular dilatation disease: identification of a candidate etiologic agent. Virol J. 2008;5:88.
2. Honkavuori KS, Shivaprasad HL, Williams BL, et al. Novel borna virus in psittacine birds with proven- tricular dilatation disease. Emerg Infect Dis. 2008;14:1883–1886.
3. Gancz AY, Kistler AL, Greninger AL, et al. Experimental induction of proventricular dilatation disease in cockatiels (Nymphicus hollandicus) inoculated with brain homogenates containing avian bornavirus 4. Virol J. 2009;6:100.
4. Gray P, Hoppes S, Suchodolski P, et al. Avian bornavirus causes proventricular dilatation disease: the isolation and propagation of avian bornavirus and the fulfillment of Koch’s postulates. Emerg Infect Dis. 2010;16(3):473-479.
5. De Kloet SR, Dorrestein GM. Presence of avian bornavirus RNA and anti-avian bornavirus antibodies in apparently healthy macaws. Avian Dis. 2009;53:568–573.
6. Lierz M, Hafez HM, Honkavuori KS, et al. Anatomical distribution of avian bornavirus in parrots, its oc- currence in clinically healthy birds and ABV-antibody detection. Avian Pathol. 2009;38:491–496.
7. Rinder M, Ackermann A, Kempf H, et al. Broad tissue and cell tropism of avian bornavirus in parrots with proventricular dilatation disease. J Virol. 2009;83:5401–5407.
8. Hoppes S, Gray P. An update on proventricular dilatation disease. Proc Conf Euro Assoc Avian Vet. 2009;141–146.