Susan L. Clubb, DVM, Dipl. ABVP Avian
P.O. Box 508 Loxahatchee, FL
Carolyn Cray, PhD University of Miami
Dept of Comparative Pathology
Miami FL
Ellis Greiner, DVM, PhD, College of Veterinary Medicine
University of Florida Gainesville, FL
Kenneth S. Latimer, DVM PhD College of Veterinary Medicine
University of Georgia
Athens, GA
Session # 5050
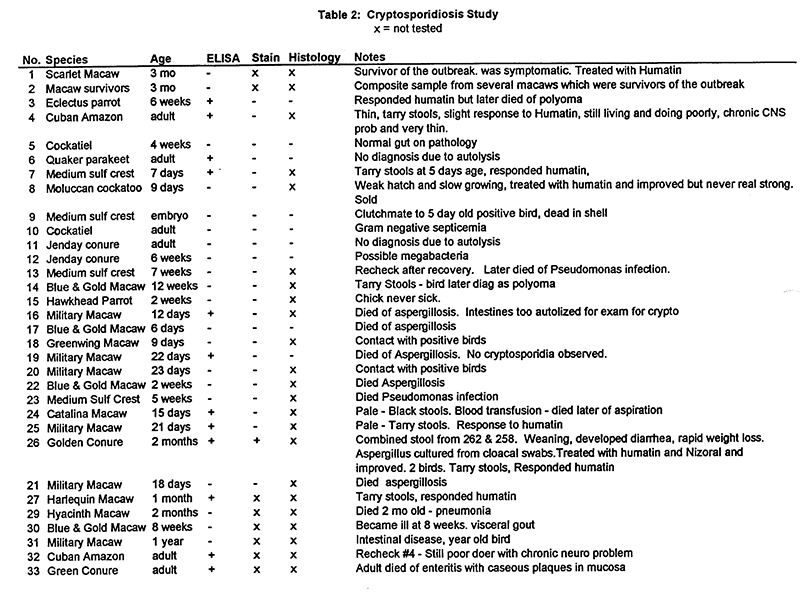
In August 1994 a disease outbreak occurred among neonatal macaws in a psittacine nursery. Seventeen (17) neonatal psittacines (age 2-28 days), 16 macaws, and an African grey parrot died in the outbreak. Three birds were euthanized for pathology and in all three cryptosporidiosis was diagnosed. Cryptosporidium was found in fecal smears stained with Kinyoun’s Modified Acid-Fast Stain from an additional 3 of 5 which were showing clinical signs. A number of macaws survived the outbreak after treatment with paromomycin.
Clinical disease occured primarily in macaw chicks between 2-4 weeks of age. (Table 1) All of the chicks were incubator hatched and handreared using a commercial handrearing formula and well water. Untreated disease course was 3 to 10 days which seemed very protracted for neonates. Affected birds were severely stunted, emaciated, pale and profoundly dehydrated (approx 10%). Their abdomens were distended due to severe serosal edema of the intestines and ascites. Serosal edema was visible through the skin and abdominal wall. Chicks exhibited melena and gut stasis. Feather development, eye opening and other developmental parameters were delayed. Terminally ill chicks became ataxic, dyspenic, weak and couldn’t stand, showing terminal clinching of the feet and muscle rigidity. Choanal and cloacal aerobic cultures on 12 birds, including three euthanized birds, were insignificant. Treatment with antibiotics (enrofloxacin, piperacillin), antifungal drugs (nystatin, ketoconazole), fluids, and intestinal stimulants (metoclopromide) was ineffective.
Three birds, 2 caninde macaws (Ara glacogularis) which were 21 and 25 days old and a 28 day old Military macaw (Ara militaris) were euthanized. On post mortem exam chicks were emaciated with little subcutaneous fat, muscle atrophy and soft bones. They had severe edema in the subserosal tissues of the duodenum and primarily the jejunum. Some had a variable degree of ascites Spleens were enlarged (a number of the birds were euthanized). Livers were somewhat enlarged and friable. In some birds, kidneys were enlarged and/or pale. Fibrin deposits were found between the pericardial sac and sternum. Some showed suffusive hemmorrhages in the calivarium,

which occasionally were visible antemortem. Some showed petechia of the wing tips and feet. Airsacs were clear. Thymuses and bursas were very small.
On histopathology, moderate to severe multifocal intestinal cryptosporidiosis was found in all three birds. The intestinal villi were blunted and occasionally fused. Numerous sperical protozoal organisms, approximately 5 micrometers in diameter were present on the epithelial surface of the intestine and colon. These organisms occasionally contained 2 to 3 banana shaped meronts and were consistent in morphology to Cryptosporidium sp.
The chicks had mild to severe diffuse heterophilic serositis with edema. The colonic serosa was severely edematous and contained mild diffuse to focally severe infiltrates of heterophils and fewer lymphocytes and plasma cells. The inflammation often extended into the adjacent muscularis and lamina propria. Similar serosal infiltrates were present on the surface of the spleen and to a lesser degree on the ventricular surface. In one bird, particles of plant material were scattered throughout the inflammatory infiltrates within the intestinal serosa.
The bursas were atrophic. Central follicular necrosis of the lymphoid elements was present. Multifocal mild heterophilic infiltrates were within crypts and the surrounding intrafollicular stroma. Bursal epithelium was hyperplastic and squamous metaplasia was observed in numerous areas.
The sheathed vessels of the spleen contained proliferations of phagocytic cells containing intracytoplasmic protein. Sections of liver contained small multiple areas of hepatocellular degeneration and necrosis.
In one bird, the military macaw, the white matter of the cerebellum contained individual cell necrosis and scattered vacuoles. Occasional mitotic cells were present within the vacuolated areas and suggested attempts at regeneration. Increased numbers of gemistocytes and glial cells were observed within the central white matter of the cerebellum. The submucosal proventricular glands contained severe diffuse vacuolization. Mild multifocal heterophilic infiltrates were present within the muscularis mucosa and extended between glandular elements. Focal mineralization and necrosis was present within the myocardium. Rare foci of myocardial degeneration also were observed.
The intestinal cryptosporidiosis was likely the cause of the intestinal lesions in the multiple chicks. Lesions in the liver, spleen, heart and thoracoabdominal serosa of the military macaw were suggestive of bacterial infection and subsequent septicemia, possibly from yolk-sac infection. The plant material within the serosal inflammation is suggestive of a intestinal perforation or post mortem contamination. Squamous metaplasia in the bursa is suggestive of vitamin A deficiency. The cause of the degenerative lesions in the cerebellum is unknown. It is similar to the vacuolar form of vitamin E deficiency-induced encephalopathy in poultry. These vitamin deficiencies may have been associated with malabsorption. Bursal lymphoid atrophy, necrosis and inflammation were commonly seen in stressed or ill birds.
Treatment
Paromomycin, a broad spectrum macrolide antibiotic has shown promise as the first drug effective for treatment of cryptosporidiosis. Paromomycin was used in this outbreak after the diagnosis of cryptosporidiosis was made and for cases diagnosed subsequent to the outbreak. Response was typically immediate and remarkable. In this outbreak paromomycin was used at 100 mg/kg orally BID for 7 days. A 250 mg capsule was mixed with 10 ml of water to make a suspension for ease of dosing.
Diagnosis
Initial diagnosis was made by histopathology and confirmed by microscopic examination of acid-fast stained fecal smears. Subsequently, a survey study was undertaken in an attempt to determine the source of cryptosporidiosis. A study was designed to contrast three diagnostic techniques: ELISA, acid fast staining and histopathology (in case of mortality). Cases selected were symptomatic birds, contact birds and randomly selected necropsy cases from the farm where the outbreak occurred. Only 2 birds (# 3 and 30) were from sites other than the original site and they were included due to melena (tarry feces). Both of these birds had polyomavirus infection. Results of this study are presented in Table 2.
Acid Fast Staining of Feces
Fresh feces were liberally smeared onto a microscope slide and air dried. Slides were stained using the Kinyoun’s Modified Acid Fast technique. Slides are examined at 100x and 400x for the presence of red non-homogeneous oocysts (3.5-5 mm) consistent with Cryptosporidium sp. These sporozoites stained darker red. Oocysts were found in low numbers.
Cryptosporidium Antigen ELISA
The ProSpecT Cryptosporidium Microplate Assay (Alexon, Inc., Sunnyvale, CA) was used to detect Crytosporidium antigen for this study. This solid phase immunoassay utilized monospecific antibodies to Cryptosporidium specific antigen (CSA). If CSA was present in the sample, it was bound to the antibody present in microplate wells. A second step anti-CSA antibody, which is conjugated to horseradish peroxidase (HRP), was added. In a positive reaction with a final incubation with a substrate (TMB), a colored reaction product was formed. This color change was quantified by use of an ELISA reader at a 450nm wavelength (Molecular Devices, Menlo Park, CA). The optical density (O.D.) of the negative control well was subtracted from the positive controlO.D. The positive control was within acceptable limits if this result was greater than 0.3. A test sample was considered positive if the normalized O.D. was greater than 0.05.
The Alexon kit has been found to detect approximately 20 nanograms/ml of CSA. The assay has been tested extensively by the company for cross reactivity with a number of fecal parasites. No reactivity was found with a number of parasitic worms as well as Giardia.
Further studies at the University of Miami laboratory have found that the presence of occult blood in the fecal samples does not create false reactions (data not shown). Less than 6% of the samples randomly submitted to the laboratory were found to be positive by this assay. With the use of a known positive submission, storage conditions were found to be critical for test detection. The limits of the assay were as follows: submission of fresh (< 48 hours old) feces or feces preserved in 10% formalin (a few drops to moisten fecal specimen) within one week of collection.
Study results
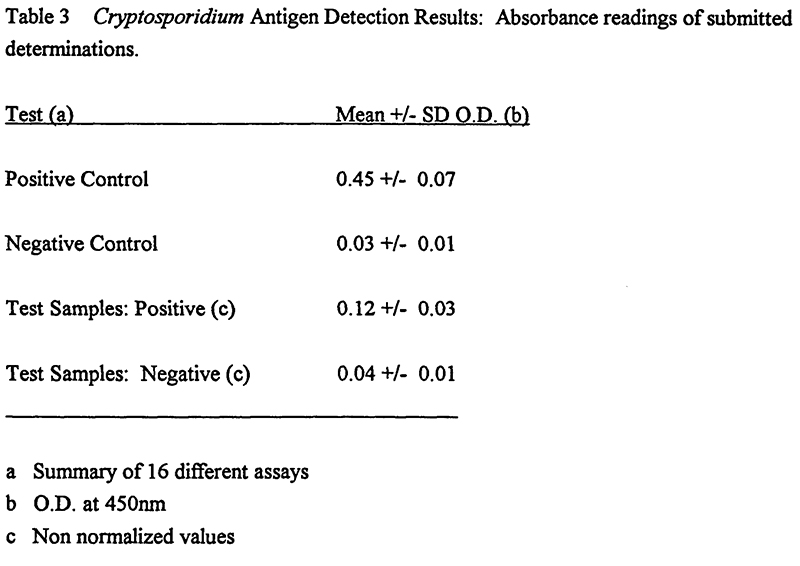
Unpreserved fecal specimens were analyzed within 36 hours of collection using the Alexon kit. Positive O.D. were within assay limits. All positive test results were observed to fall in the low range of the assay (Table 3). The normalized (i,e, negative control O.D. subtracted) test values ranged from 0.06-0.12. Non-normalized values were statistically significant (vs. negative control, p<0.0001).
Discussion
Tyzzer was the first to report cryptosporidia in birds in 1920. Two valid species of Cryptosporidium are now recognized in avian hosts. Cryptosproidium meleagridis is found in turkeys and C. baileyi is found in chickens. Cryptosporidiosis in birds causes enteritis, respiratory disease or renal disease. Usually only one form of disease is present. In naturally occuring cases, Cryptosporidia have been reported to develop in many tissues including conjunctiva, nasopharynx, larynx, trachea, bronchi, airsacs, proventriculus, small intestine, large intestine, ceca, cloaca, bursa of Fabricius, kidneys and urinary tract. Fully sporulated oocysts are passed in the feces. Following ingestion of oocysts, the primary sites of development are the Bursa of Fabricius and the cloaca. Transmission studies using various avian cryptosporidia have failed to produce infection in 10 species of mammals, which indicates that zoonotic potential is not present.1
The life cycle of C. baileyi includes asexual stages that recycle, autoinfective oocysts and fully sporulated fecal oocysts. Age-related resistance to clinical disease and the ability to clear C. baileyi has been demonstrated in chickens. Chickens were most susceptible to infection and disease developed between 2 and 14 days of age.1,2
Enteritis associated with cryptosporidia has been reported in budgerigars, cockatiels and lovebirds. An outbreak of enteric cryptosporidiosis was seen in 7 to 14 day old lovebirds (Agapornis sp.).3 Almost all chicks in the aviary died within a month of hatching. At necropsy, the chicks were dehydrated and had dilated, yellow fluid filled intestinal tracts. Cryptosporidia were seen in the jejunum and ileum and associated with villus atrophy and villus fusion. No parasites were seen in the respiratory tract. The high mortality and clinical manifestations of the disease outbreak seen in lovebirds is similar to that seen only previously in bobwhite quail.1 Postmortem findings may include dilated intestines containing yellowish fluid and blunting, fusion, and atrophy of intestinal villi. Kidneys may be enlarged and pale. Proventricular lesions have been described.4
Respiratory cryptosporidiosis was observed in a budgerigar (Melopsittacus undulatus). The bird was found dead, and no clinical signs were observed. Grossly, the lungs were congested and there was excess mucus in the trachea. Microscopic lesions were subacute tracheitis, fibrinous airsacculitis, and pulmonary congestion. Cryptosporidia were seen only in the trachea.1
Cryptosporidia have been seen in the small intestines of a budgerigar that had diarrhea. The lamina propria of the small intestine was infiltrated with sheets of plasma cells and lymphocytes, and cryptosporidia were present on the distal portions of the villi.
Small intestinal cryptosporidiosis has been seen in cockatiels (Nymphicus hollandicus). Diarrhea was reported in both cases. In one study, the small intestine was inflitrated with lymphocytes and plasma cells. In the other study no inflammatory response was present.1
Cryptosporidia have been observed as incidental findings in the cloacal coprodeum of a red-lored parrot (Amazona autumnalis) and in the feces of a 7 year old macaw (Ara sp.)4,5
Control
Historically chemoprophylaxis or treatment of cryptosporidiosis has been unrewarding and control has been aimed at environmental sanitation. Several commonly used disinfectants have been evaluated for the ability to kill C. baileyi oocysts in an excystation assay. None of the disinfectants was effective at recommended concentration. Sodium hypochlorite (household bleach, 5.25%) used at 50:50 ration with water was somewhat effective. Improved sanitation, cleaning with bleach and exposing items to direct sunlight for three days may be somewhat effective in reducing incidence of infection.1,2
Paromomycin (Humatin, Parke Davis, Morris Plains, NJ) has an antibacterial action which closely parallels that of neomycin. It is poorly absorbed after oral administration, with almost 100% of the drug recoverable in the stool. It is indicated for acute and chronic intestinal amebiasis. The use of paromomycin may result in overgrowth of nonsusceptible organisms including fungi. The drug should be used with caution in individualals with ulcerative lesions of the bowel to avoid renal toxicity.7
Paromomycin has been reported to be highly effective, non-toxic drug, against diarrhea in dairy and beef calves associated with Cryptosporidium. Calves treated daily with 100 mg/kg body weight were 100% protected when challenged with cryptosporidial organisms. USDA and Walter Reed Army Institute of Research have applied for a patent to use paromomycin against Cryptosporidium in animals. Currently, there is no approved treatment for this parasite.8
Cryptosporidiosis can be transmitted through ingestion or inhalation of sporulated oocysts. The life cycle is direct. Cryptosporidium undergoes endogenous sporulation resulting in autoinfection in the parasitized host. As few as 100 oocysts can induce severe enteritis and diarrhea in experimentally infected bob-white quail in the company of reovirus.2
The small size of Cryptosporidium oocysts and low shedding rate make diagnosis difficult. Staining techniques include Kinyoun’s modification of the Ziehl-Neelsen acid-fast stain, iodine solution, and Giemsa stain. Microscopy techniques include brightfield, phase-contrast and Normarski interference-contrast. Auramine 0 staining and fluorescent microscopy was used in a study in which fecal smears from 113 avian necropsy accessions representing 436 birds were stained and examine for Cryptosporidium oocysts. In this survey 18.2% (6/33) of the psittacines sampled were Cryptosporidium positive including a macaw and a budgerigar.9
In some cases Cryptosporidium is considered a primary pathogen; however, in most situations it is considered to cause severe infections only in immunocompromised hosts. Suggestive of the opportunistic nature of Cryptosporidium was the detection of the parasite in four cockatoos with psittacine beak and feather disease virus.10
In this outbreak, severe intestinal cryptosporidiosis was associated with high mortality in neonatal macaws, between the ages of 2 and 28 days. After diagnosis was made, treatment with paromomycin at 100 mg/kg orally, BID for 7 days, stopped the mortality except in those birds which were already extremely debilitated. Subsequent to the outbreak, chicks showing clinical signs - melena, pallor, depression - were immediately started on paromomycin while ELISA tests were pending. Response was typically evident within 24 hours. The Alexon ELISA assay for
Cryptosporidium Specific Antigen was found to be a very specific and sensitive test for cryptosporidiosis in neonatal psittacines and correlated very well with clinical signs and response to treatment.11
References
1. Lindsay, David S., and Byron L. Blagburn, Cryptosporidiosis in birds, in Cryptosporidiosis of Man and Animals, J.P. Dubey, C.A. Speer and R. Fayer, CRC Press, Boca Raton, FL
2. Goodwin, MA, Cryptosporidiosis in birds - a review. Avian Pathol. 18:365-384, 1989.
3. Belton, DJ, Powell IB, Cryptosporidiosis in lovebirds (Agapornis sp.) New Zealand Vet, J., 35:15, 1987.
4. Greiner, Ellis, C., B.W. Ritchie., Parasites, In Avian Medicine, Principles and application, Ritchie, Harrison and Harrison, Wingers Publishing, Lake Worth FL 1005-1029, 1994.
5. Doster, A.R., and E.A. Mahaffey, Cryptosporidia in the cloacal coprodeum of red-lored parrots (Amazona autumnalis). Avian Dis. 23:654-661. 1979.
6. Ley, David, H., Avian Cryptosporidiosis - An Emerging Disease, Proceedings, First International Conference, Zoological and Avian Medicine, 299-303, 1987.
7. Package insert - Humatin, Parke Davis, Div of Warner-Lambert Co, Morris Plains, NJ.
8. Florida Veterinary Scene- Veterinary Medicine Newsletter , University of Florida, Cooperative Extension Service, Institute of Food and Agricultural Sciences, Vol 4, No. 7, August 1995.
9. Ley D.H. et al: Cryptosporidium positive rates at avian necropsy accessions determined by examination of auramine O-stained fecal smears. Avian Dis 32:108-113, 1988.
10. Latimer, KS et al, Cryptosporidiosis in four cockatoos with psittacine beak and feather disease. J. Am Vet Med Assoc., 200:107-110, 1992.
11. Mohan R. Evaluation of immunofluorescent and ELISA tests to detect giardia and cryptosporidium in birds. Proc Annu Conf Assoc Avian Vet 1993;62-64.
