Susan L. Clubb, DVM, Dipl ABVP (Avian), Michael M. Garner, DVM, Dipl ACVP, Carolyn Cray, PhD, Kris Arheart, and Michelle Goodman
Session #680
Affiliation: From The Rainforest Clinic for Birds & Exotics, PO Box 508, Loxahatchee, FL 33470, USA (Clubb), Northwest Zoopath, 654 West Main, Monroe, WA 98272, USA (Garner), University of Miami, Division of Comparative Pathology and Avian and Wildlife Laboratory, University of Miami School of Medicine, Miami, FL 33101, USA (Cray), Department of Epidemiology and Public Health, University of Miami School of Medicine (Arheart), and Kaytee Products Inc, 521 Clay Street, PO Box 230 Chilton, WI 53014, USA (Goodman).
African grey parrots (Psittacus erithacus) are well known for their beauty, intelligence, and ability to mimic human language. They are also well known for feather damaging behavior (FDB). Self-destructive behaviors in psittacine birds, such as feather picking or chewing and soft tissue mutilation, are common, often multifactorial problems. While generally believed to be behavioral in etiology, FDB still has not been fully explored from a diagnostic point of view. Establishment of a definitive etiologic diagnosis and effective therapeutic plan is challenging.
The role of hypothyroidism is often mentioned in association with FDB but is controversial. In a previous study (Clubb et al), diagnostic data indicated an association between feather damaging behavior and low plasma T4 levels.1
Skin biopsies are often recommended as an important diagnostic tool in self-mutilation, plucking, or feather destruction cases. Examination of only the affected site, however, provides a limited amount of information upon which to base a diagnosis. Most sites of active plucking or mutilation will exhibit inflammation, even if only associated with self-induced trauma. Examination of an unaffected section of skin allows the pathologist to detect generalized lesions that may be interpreted as inflammatory skin disease. This is very useful in differentiating cases of psychogenic feather damage from birds that have generalized inflammatory skin disease due to allergies or other disorders.1,2
This is a preliminary report from an ongoing study in which the authors attempted to review as many diagnostic modalities as possible in a population of African grey parrots with similar husbandry standards and exhibiting FDB. In order to provide sufficient samples for statistical analysis, normal birds from outside this population were needed.
Materials and Methods
A database was established in order to collect uniform data, which could be used to establish a baseline on feather damage in this collection. The primary collection used in this study consisted of 24 adult (13 females and 9 males), wild-caught African grey parrots, housed in a single facility and fed a commercial diet ad libitum. Precise ages are unknown, but they had resided in this collection for at least 10 years. These birds had normal baseline complete blood counts (CBC) and blood chemistry profiles for the previous 3 years. As quantities of normal birds were insufficient for statistical analysis, an additional 6 unaffected (normal plumage) birds were included from a zoo collection for the blood values portion of the study.
For initial blood collection, each bird was captured and a blood sample collected from the jugular vein with manual restraint, within a maximum of 30 seconds, for hormonal assays (pre T4, corticosterone, serotonin) and CBCs. In each case, sampling was initiated between 9:00 and 10:00 am to avoid the effects of diurnal variation. Blood was placed in pediatric-sized lithium heparin plasma separator tubes (VWR, West Chester, PA, USA) and separated immediately. Two peripheral blood smears were prepared immediately on full-length glass slides. Thyroid stimulating hormone (0.5 IU; TSH, Thyrotropic hormone from Bovine Pituitary, Sigma 10 IU/vial, Lot 102K1090, Sigma- Aldrich, St Louis MO, USA) was administered intramuscularly into the pectoral muscle.3 The second blood sample was collected 6 hours afterward for a determination of the post TSH T4 level. All samples were analyzed at the University of Miami Avian and Wildlife Laboratory except initial CBCs (Marshfield Laboratory, Marshfield, WI, USA).
Total WBC was determined by the Unopette method. WBC differential was performed manually after staining smears in Diff Quik (Southeast Vet Lab, Miami, FL, USA). The remaining blood was centrifuged and plasma stored at -20oC until further testing was completed. This testing included protein electrophoresis, Aspergillus antibody and antigen serology, Candida antibody serology, thyroxine, corticosterone, histamine, and serotonin. Protein electrophoresis was completed as previously described.4 Values were compared as percent of total protein. Aspergillus antibody and antigen serological testing was performed by ELISA as described.5 Values were calculated from the equation (absorbance of test sample/mean absorbance of negative samples). Candida serology was performed using an adaptation of a commercial kit (Candida IgG, ALPCO Diagnostics, Windham, NH, USA). The kit procedure was followed and anti-chicken IgG (Sigma) was substituted at the conjugate stage. The final result was calculated in the same format as the Aspergillus test. Thyroxine and corticosterone were determined by radioimmunoassay (Diagnostic Products Corporation, Los Angeles, CA, USA). Serotonin and histamine were quantitated by ELISA assays also purchased from ALPCO Diagnostics. All samples were analyzed in duplicate. Fecal smears of each pair of birds were evaluated by Gram stain (Clubb).
Two weeks later each bird was anesthetized for paired skin/feather biopsies. Two samples were collected from affected areas and 2 from unaffected areas. Growing immature feathers were sampled with a full thickness section of skin, which included the entire feather follicle. A growing immature feather was selected in an area where the bird was affected (plucking or other feather damaging behavior) and another was collected in an area which was unaffected, typically the head or neck. Samples were fixed in 10% buffered formalin for histopathology. The formalin fixed specimens were sectioned routinely and examined at 3 different levels. Each bird was photographed and the patterns of feather damage were described for future comparison by estimation of amount and type of feather damage in each major body area. Blood was again collected for plasma zinc levels, psittacine beak and feather disease PCR, and polyoma serology (Infectious Diseases Laboratory, University of Georgia, Athens, GA, USA). Plasma zinc levels were determined by Flame ASS.
Data were analyzed as percentages and absolute numbers. Log transformation was used on the data and it was analyzed by analysis of variance. One way analysis of variance (ANOVA) was used for the data that were measured once. For variables measured before and after treatment, a repeated measures analysis of variance with planned comparisons was used to test the significance of pre-post differences within groups. The 0.05 significance level was used to determine statistical significance. The Statistical Analysis System (SAS) (SAS, Incorporated, Cary, NC, USA) was used for data management and analysis.
Results
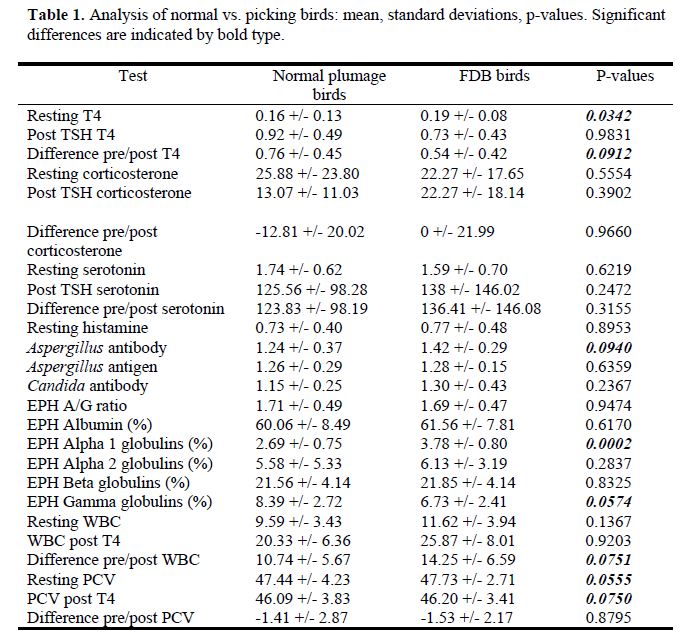
A summary of results are presented in Table 1.
The following comparisons are made between pre- and post- sampling values, and reflect changes caused by TSH administration or handling (these p values are not contained in the chart above).
- Differences for both the normal group and for the FDB group between post vs. pre T4 levels were significant (p<0.0001)
- Differences for both the normal group and for the FDB group between post vs. pre serotonin levels were significant (p<0.0001)
- Differences for both the normal group and for the FDB group between post vs. pre WBC were significant (p<0.0001)
- Differences for both the normal group and for the FDB group between post vs. pre PCV were significant (p=0.0296 for normals; p=0.0050 for FDBs)
- Differences for both the normal group and for the FDB group between post vs. pre Heterophils were significant (by % and absolute #s)
- P=0.0004 for normals; p<0.0001 for FDBs
- Differences for both the normal group and for the FDB group between post vs. pre lymphocytes were significant, by % and absolute # (p<0.0001)
All birds in the study were negative by PCR for circovirus I (PBFD). Five of 24 birds had antibody titers to avian polyomavirus. Two birds had antibody titers of 1:20, one was 1:80, one 1:160, and one 1:1280. A titer of 1:20 is considered weak positive. Birds recovering from recent avian polyomavirus infection typically have antibody titers of greater than 1:160 (interpretation provided by Infectious Diseases Laboratory). There have been no known cases of polyomavirus infection or confirmed cases in neonates in this collection in the last 5 years. Plasma zinc levels were all within the reference range. No significant abnormalities were found on fecal Gram stain.
Little variation in lesions existed between the biopsies from affected areas vs unaffected areas in FDB birds. The most common lesions in both affected and non-affected areas were dermal scarring and perivascular inflammatory cell cuffing. Scarring was noted in 71% of the affected locations and 66% of the unaffected locations. Inflammation was noted in 83% of the affected locations and 71% of the unaffected locations.
Resting T4 values were significantly lower in birds with FDB than in normal birds (p=0.03). The response to TSH stimulation was lower in birds with FBD than in normal plumage birds (p=0.09).
Normal birds had a lower antibody to Aspergillus species than did birds with FDB (p=0.09). Normal birds had lower alpha 1 globulins (non-specific indicator of inflammation) than FDB birds (p=0.0002). Normals had lower gamma globulins (indicator of humoral immune stimulation/inflammation) than FDB birds (p=0.06).
With a TSH injection/blood draw, normal birds demonstrated a lower WBC change than did FDB birds (p=0.08). Resting PCV values and post-TSH injection PCV values were slightly higher in FDB birds than in normal birds.
Discussion
Perivascular inflammation was found in 83% of affected feather biopsy sites and 71% of non-affected sites. These findings likely are not significantly different and indicate that most of the affected birds likely had some form of inflammatory skin disease, such as hypersensitivity or a cutaneous manifestation of a systemic inflammatory event.
There was no significant difference between FDB and normal birds in resting and post TSH/handling corticosterone, or in serotonin levels. There was no significant difference between FDB and normal birds in resting histamine levels.
Hypothyroidism has been loosely associated with feather destruction.1,2 The biological functions of the thyroid gland in birds include normal molting and development of the plumage. Feather abnormalities that have been associated with hypothyroidism in poultry include fringed feathers, loss of barbules resulting in a silky feeling, and hypopigmentation of colored feathers.6 The diagnosis of hypothyroidism is based on characteristic clinical signs coupled with the failure to respond to provocative TSH-stimulation testing. Techniques for provocative testing using TSH have been described.3,7-9
A previous study of multiple species including 7 African grey parrots revealed relatively low rates of inflammatory skin disease (1:7, or 14.3%) but high incidence of low resting thyroxine levels (100%). The reference ranges for resting T4 used for this comparison were developed by the Comparative Pathology Division of the University of Miami using random samples submitted by practitioners who indicated the birds were clinically normal. The reference range for plasma thyroxine in 120 clinically normal African grey parrots was 0.3-2.1.
The low incidence of inflammatory skin disease previously reported may be a function of being more acute cases involving younger birds with FDB of shorter duration.1 The affected birds in this study had been exhibiting FDB for many years, which may have resulted in, or resulted from, more extensive, chronic systemic inflammatory disease.
Conclusion
Anecdotal associations between FDB and Aspergillus infection have been made previously. This is the first documentation of a significant difference in antibody titers between normal and African grey parrots with FDB. Additional study is needed with repeat testing of the affected birds to further dissect this apparent correlation.
These data showed a strong correlation of FDB to inflammatory skin disease as well as reduced or sub-optimal thyroid gland function in this population of African grey parrots. The difference in response to TSH stimulation was significantly lower in FDB birds than in normal birds. Further studies will be conducted to increase sample size for this project as well as to examine paired samples.
Acknowledgments: Study sponsored by Kaytee Avian Foundation.
References
1. Clubb SL, Garner MM, Cray C. Detection of inflammatory skin disease in psittacine birds using paired skin biopsies. Proc Annu Conf Assoc Avian Vet. 2002; 193-193.
2. Garner MM. Avian noninfectious skin disorders. Proc Annu Conf Assoc Avian Vet: Avian Specialty Advanced Program. 2003;21-23.
3. Zenoble RD, Kemppainen RJ, Young DW, Clubb SL. Endocrine response of healthy parrots to ACTH and thyroid stimulating hormone. J Am Vet Med Assoc. 1985;187:1116-1118.
4. Cray C, Tatum LM. Applications of protein electrophoresis in avian diagnostics. J Avian Med Surg. 1998;12:4-10.
5. Cray C, Zielezienski K. Application of aspergillosis antigen testing to the diagnosis of aspergillosis. Proc Annu Conf Assoc Avian Vet. 1997:219.
6. Merryman JI, Buckles EL. The avian thyroid gland. Part two: a review of function and pathophysiology. J Avian Med Surg. 1998;12:238-242.
7. Lothrop CD. Thyrotropin stimulation test for evaluation of thyroid function in psittacine birds. J Am Vet Med Assoc. 1985;186:47-48.
8. Oglesbee BL. Hypothyroidism in a scarlet macaw. J Am Vet Med Assoc. 1992;201(10):1599-1601.
9. Greenacre C, Young DW, Behrend EN, Wilson GH. Validation of a novel high-sensitivity radioimmunoassay procedure for measurement of total thyroxine concentration in psittacine birds and snakes. Am J Vet Res. 2001;62(11):1750-1754.